UK MHRA Medical Device & IVD Registration: Step-by-Step Guide

Team Notified Body (NB) Position Paper: Medical Device Lifetime
December 18, 2023
MDCG 2021-27 Updated: Q&A on MDR/IVDR Articles 13 (Importers) & 14 (Distributors)
December 20, 2023- Who is allowed to submit UK registrations?
- How do I register for an online MHRA account?
- How do I log-in to my MHRA account?
- How do UK Manufacturers start a new application?
- What device information is required?
- How many device families can be in one application?
- Can I save a draft of an application and edit it later?
- How do I submit the application?
- What is the MHRA application fee?
- How do I pay the registration fee?
- How long does it take to register?
- Can I make changes to the approved registration?
- How long is the registration valid?
- Can devices with expired CE Certificates under EU Regulation 2023/607 be registered?
- Do custom-made devices need to be registered?
- Is there a public registration database?
All medical devices and IVDs must be registered with the MHRA before they can be placed on the UK market. Below are the steps for UK-based manufacturers to create an MHRA account and register their devices.
Who is allowed to submit UK registrations?
The MHRA medical device registration can only be completed by companies located in the UK: either UK manufacturers or a foreign manufacturer’s UK Responsible Person (UKRP).
Further, it must be completed in the MHRA’s online database, called ‘DORS’. It is a proprietary account that the UK company will create to submit and maintain registrations.
Below are the steps for UK-based manufacturers to create a DORS account and register their devices. The steps are based on MHRA user guides:
- Account Management Guide (42 pages)
- Device Registration Guide (99 pages)
The process is similar, though slightly different, when a UK Responsible Person submits the registration on behalf of the non-UK manufacturer.

How do I register for an online MHRA account?
To register for an MHRA ‘DORS’ database account, click: HERE
Once you click on the link, you must provide basic details about:
- Type of organization, including company name, address, and role (UK Manufacturer, UK Responsible Person, or Northern Ireland Authorized Representative)
- Account user details, including name, email address, and telephone number
- Primary reason for the account: 1) applications related to medical devices (and IVDs) or 2) submissions related to medicines and e-cigarettes
After you hit submit, the MHRA will send two automated emails:
- “Account creation – outcome”, which confirms the account has been created and provides links to the user guides and tutorials.
- “MHRA Portal account creation”, which will contain your user name (usually firstname.lastname) and a temporary password.
- The temporary password will expire within 7 days, so companies should not wait to log-in.
- You will be required to create a new password the first time you log-in.
How do I log-in to my MHRA account?
After you have received your username and temporary password, you can log-in: HERE
Select your organization’s purpose (e.g., Medical Devices) and accept the MHRA’s cookie policy to access the main database.
How do UK Manufacturers start a new application?
Upon logging into the MHRA DORS system, you will see the below home page. To open a new registration application, select “Enter”.
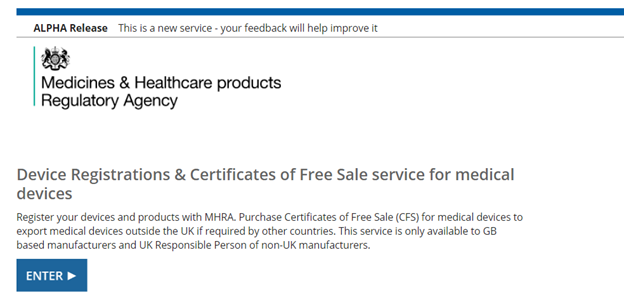
On the next page, click on your company name, which will be located under “Device Registration & Certificates/Your Organisation”:
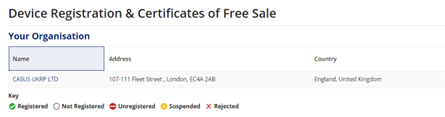
On the next page, select “Add Devices” from the top right corner of the screen.
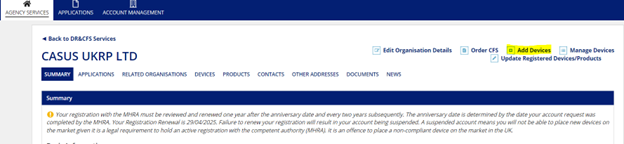
This will take you to the registration application.
What device information is required?
The following information is required in the medical device registration request.
Device Details
- GMDN code
- See Resource Page: How to find GMDN Codes for Free
- Device type: Medical device, IVD, Active Implantable or System/Procedure Pack (S/PP)
- Regulation leveraged (E.g., UK MDR 2002, MDD, IVDD, MDR, IVDR, etc.)
- Devices must be CE and/or UKCA marked* to lawfully be placed onto the market
- Custom-made or not
- Risk classification
- Sterility (and type, if sterile)
- Active/non-active
- Implantable/non-implantable
- Single-use/reusable (and how many times it can be reused, if applicable)
- Administer and/or remove medicinal product
*From a regulatory standpoint, the UK is made up of two territories:
- Great Britain (England, Scotland, Wales) is subject to the UK MDR 2002 requirements. This means devices may be either UKCA or CE Marked; and non-UK companies must appoint a UKRP.
- Northern Ireland, the fourth UK country, does not and will not recognize UKCA Marking. Due to Brexit negotiations, Northern Ireland will continue to be part of the European Single Market and require CE Marking.
Evidence of Conformity Assessment
- The type of evidence will vary based on the device type (e.g., medical device, system/procedure pack) and regulation leveraged (e.g., UK MDR 2002, MDD, IVDR)
- General Medical Devices, AIMD & IVDs:
- Declaration of conformity (self-certified medical devices and IVDs)
- Notified Body/Approved Body Certificates (higher risk medical devices and IVD
- Custom-made Devices:
- Custom-made device statement
- System/Procedure Pack:
- S/PP Declaration
- General Medical Devices, AIMD & IVDs:
Device name and model/product codes
- Medical Device Name and the Device Identifier (model/version and/or catalogue/reference number)
- You must enter the device name, plus at least one mode/version number or one catalogue/reference number, per device
- For a high volume of device models/catalogue numbers, use the MHRA’s “Bulk Upload Form” to save time
- All other device details (e.g., UDI information, quantity per package, etc.) are optional; you may enter them if you wish, but you are not obligated to do so
- Confirmation if that particular device will be placed on the Great Britain and/or Northern Ireland market
- NOTE: Northern Ireland does not recognize UKCA Marking; therefore, if you register leveraging your UKCA Marking, you should only place your devices onto Great Britain (England, Scotland, Wales). Northern Ireland, the fourth UK country, requires CE Marking.
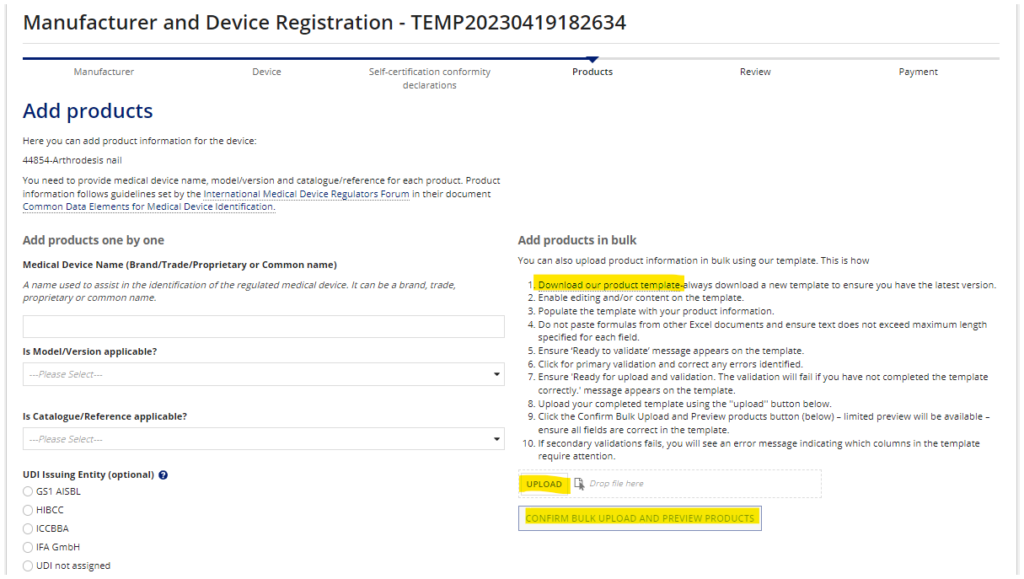
What is the purpose of the “Bulk Upload” spreadsheet?
The MHRA provides a bulk upload form (Excel Spreadsheet) that can be used if you are uploading a large number of device models. It generally saves time if you have more than 5 models to enter.
The form can be downloaded from the “Add Products” page within the registration application.
NOTE: the form is occasionally updated by the MHRA. It will not upload correctly. Further, there are different versions of the form, depending on the type of device being registered in that specific application. Therefore, download it from the MHRA application each time to avoid using an outdated or incorrect form.
How to fill out the Bulk Upload spreadsheet
The following columns are mandatory:
- Medical Device Name
- Once you enter information in this field, all other required fields will be highlighted in red, which helps lets you know what you need to fill in to complete the form
- Is Model/Version applicable (yes/no) or Is Catalogue/reference applicable (yes/no)
- You only need to mark “yes” in one of these columns
- For whichever column you mark “yes”, you also need to complete the related column with either Model/Version names/numbers or Catalogue/Reference names/numbers
- Product Status – will the product be on the GB and/or NI Market
- You must scroll to the right, toward the end of the spreadsheet to find this column
Once you have completed all of the required columns, click “Validate” at the top of the spreadsheet. The upload will fail if you do not click “Validate” before uploading.
When the spreadsheet is validated, upload it to the “Add Products” page. Then click the “Confirm Bulk Upload and Preview Products” button. You may now scroll to the bottom of the page to review all devices added into the registration.
How many device families can be in one application?
You can include up to 100 different registrations within a single application. Between the 100 registrations, you can include a maximum of 20,000 products/models.
If you exceed 100 registrations, then you must submit a separate application, and pay a separate fee, for the additional devices.
If you exceed the 20,000 products/models per application, you can add them to the appropriate device registration after the MHRA approves the application in its entirety. There is no additional fee to do this.
What is considered a single device family/application, for UK registration purposes?
To be included in a single registration, devices must share the same GMDN code and the same device characteristics (e.g., sterility, classification, single-use versus reusable, etc.). If any of these are different, then a separate registration is required for those products. For example, if all characteristics are the same, except one set of devices is provided sterile and the other is provided non-sterile, then this will result in two registrations (within the 100 allowed in a single application).
How do I add multiple device applications into a single registration?
Once you have entered the information for the first device/device family, you will have the option to “Add Another Device”.
Once you click on this button, a new application will appear. Continue this same process until you have entered up to 100 device families.

Can I save a draft of an application and edit it later?
Yes, you will have the option to “Save & Exit” at any point in the application process.
Once an application is drafted, it will be available within the “Applications” tab on the top bar of the platform. As noted in the below screenshot, draft applications will be automatically deleted if no further action is taken within the application for 90 days.
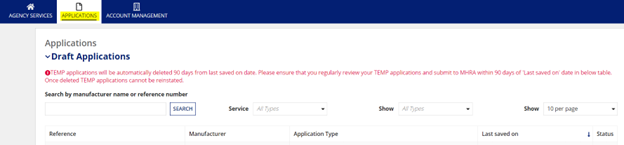
How do I submit the application?
Once you enter information for all devices, you will have the opportunity to review the information before submitting it. If needed, you can click “edit device” to fix any errors. Note that certain device edits will require you to re-enter information on some pages, including the model names.
If all the device details look accurate, then you can agree to the terms and conditions at the bottom of the page and proceed to the payment page.
What is the MHRA application fee?
The fee for one registration submission is £240.
As noted above, the application can include up to 100 registrations. If all are submitted within the same application, at the same time, only one fee will apply. However, if you submit registrations at different times and in different applications, multiple fees will apply.
How do I pay the registration fee?
First, make sure that the payment method’s address is included in your DORS account. If the billing address is the same as the original address that was entered to create the account, then you may proceed to the payment page.
If it is not, then “save & exit” the application, and edit your organization details to add a payment billing address.
Go into your organization details, select “Other Addresses” and then “Manage Other Addresses”. You will then be taken to a screen where you can add a new address. The billing address does not have to be in the UK.
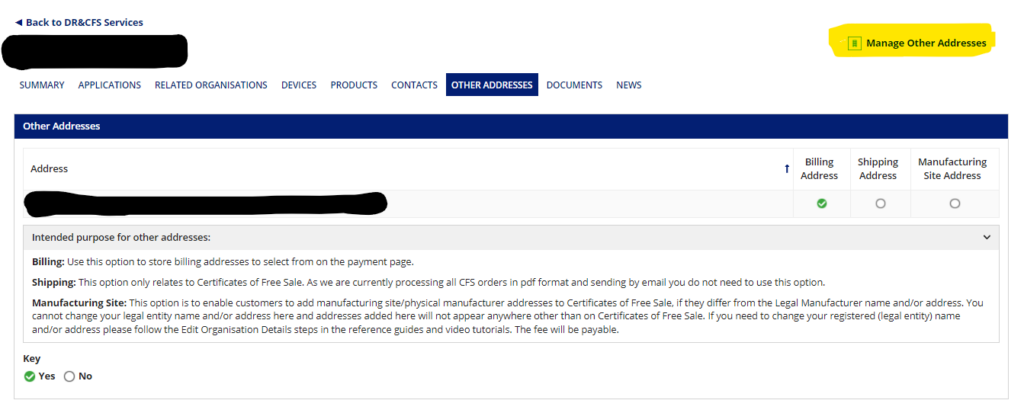
Once this is done, you may open the device application back up and proceed to the payment page.
You can pay via credit card or bank transfer.
After the payment and application are submitted, you will receive two confirmation emails from the MHRA: (1) confirmation of payment, and (2) confirmation of application submission.
How long does it take to register?
The MHRA quotes 5 days to review and process medical device registrations.
This timeframe may be shorter for registrations that have only 1-2 device families, or for well-established products. However, if an application has a larger number of device families or is for a more sensitive product (e.g., COVID-19 test), it may be longer.
In some cases, the MHRA may request additional information during their review. This may occur if they see any issues with the details included in the application.
Once the MHRA has assessed the registration request, they will issue the approval or rejection via email. If the application is approved, they will also issue a registration confirmation letter and publish the device information in their public database, PARD.
Can I make changes to the approved registration?
After registration, you can make the following changes at no additional charge:
- add additional models
- update the conformity assessment procedure (e.g., renewed CE Certificate)
- update the GMDN cod, only if the previous GMDN code was obsoleted
- change the contact person for the organization
However, you cannot edit the following:
- device details (e.g., if the device is now supplied sterile; or if the device has transitioned from MDD to MDR CE Marking)
- the manufacturer’s name or address
If these types of details need to be altered, then a new registration is required.
How to add models or update the conformity assessment procedure
To add additional models or update the evidence of conformity assessment, click “Enter” on the homepage (same as the first step in creating a new application). Then select “Manage Devices” from the top bar options. Once you select this field, you will be able to view all registered devices.
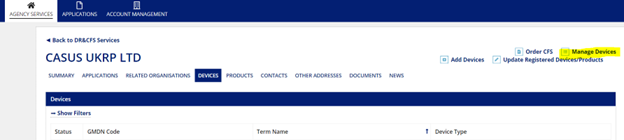
Within “Manage Devices”, scroll to the bottom of the page to view all devices. Or, if you have a high number of devices/GMDN codes, you can use their search features to filter to the device you are looking to change.
Select the registration that you would like to update. On the following screen, you will have options to both add/remove devices and to update the conformity assessment procedure.
There is no fee to update the evidence of conformity assessment or to add new models to an existing registration. Additionally, the changes are auto-approved, i.e., do not require further review by the MHRA before going into effect.
How do I change organization details?
To update the organization details, select “Enter” on the DORS home screen. Then, select “Edit Organisation Details” from the menu options in the top right corner.
All organization changes will be reviewed and approved by the MHRA, and the £240 registration fee must be paid.
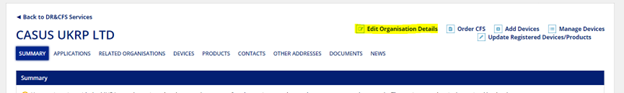
For additional details on the registration modification process, please refer to the MHRA’s Devices Reference Guide section “Updating Registrations”, which starts on page 47.
How long is the registration valid?
Registrations must be renewed one year after the initial registration. Then, every two years after that. There is no fee for the renewal.
The MHRA will send email reminders for the renewal starting three (3) months in advance of the expiration date. The renewal process is simple and requires confirmation that no device details have changed.
Can devices with expired CE Certificates under EU Regulation 2023/607 be registered?
Yes, the MHRA confirmed that expired directive CE certificates will continue to be accepted in Great Britain, if the device meets the criteria established in EU Regulation 607/2023 for the MDR extension.
The validity of these certificates are extended to:
- 31 December 2027: Implantable class IIb and class III devices
- 31 December 2028: Non-implantable class IIb, class IIa, class Im, Is and Ir devices
When uploading expired certificates for new applications, UK Manufacturers can follow the information in this MHRA guidance. The applicable steps are included in the following sections:
- Scenario 2 CE certificate expired prior to 20 March 2023 – Device/s NOT yet registered with MHRA (page 5)
- Scenario 4 CE certificate due to expire on or after 20 March 2023 – NOT yet registered with MHRA (page 7)
As noted in the guidance, a template letter must be uploaded with the evidence of conformity, attesting that the device(s) are eligible for the extension under the MDR. The template letter can be found on the MHRA’s website: HERE
Do custom-made devices need to be registered?
Yes, all custom-made devices must be registered before they are placed on the market. The registration process is the same as for non-custom-made devices.
See page 13 of the device registration guide for more information regarding how to submit the Custom-Made Statement as evidence of conformity assessment.
Is there a public registration database?
Yes, there is a public registration database: Public Access Registration Database (PARD)
The database includes device GMDN code and term, manufacturer name and address, and if applicable, the UK Responsible Person’s name and address. Full device details and model numbers are not included in PARD.
The database is searchable by manufacturer name and GMDN code name or term.


