EU Commission: New Dashboard Monitoring Availability of Medical Devices on the EU Market

MDCG 2023-7: Clinical Investigation Exemptions & Claiming Equivalence
January 4, 2024
UK MHRA: 2024-2025 Roadmap of Future Regulatory Framework
January 9, 2024The European Commission released a new dashboard, available: HERE
Purpose of the Dashboard
The EU Commission has been conducting a study on the availability of medical devices since December 2022, which will run through December 2025. This survey is being conducted due to the MDR/IVDR transition activities, and concern regarding the potential shortage of medical devices in Europe.
The study gathers information from various stakeholders, such as Notified Bodies, manufacturers, and European Authorized Representatives.
As part of this, a dashboard has been developed, which provides an overview of the survey data.
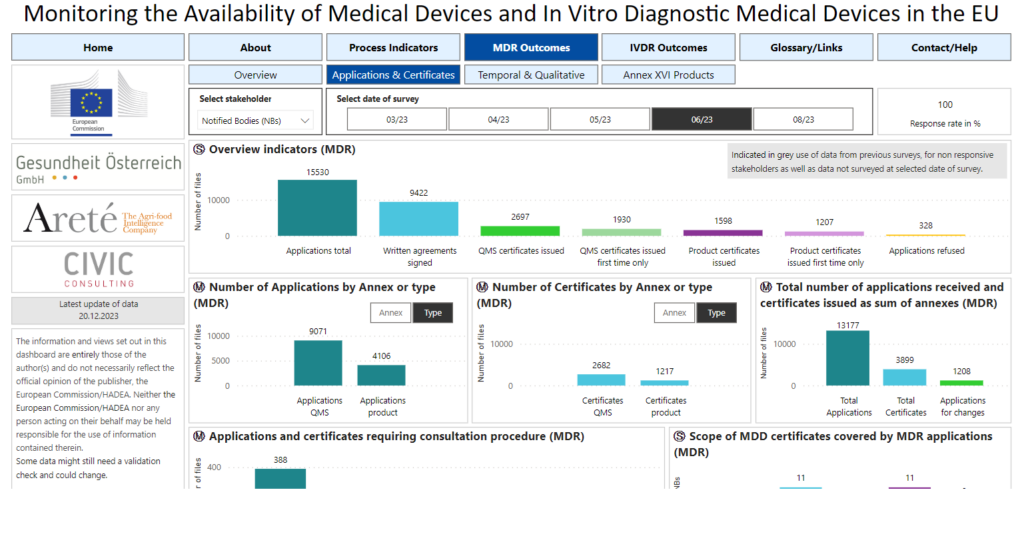
The dashboard includes information such as:
- Number of MDR/IVDR certificates in process versus issued
- How many written agreements have been signed between manufacturers and Notified Bodies
- Timeframes experienced from the point of submitting the Notified Body application, to the point of having a written agreement (important for legacy device manufacturers)
- Completeness of applications submitted by manufacturers, and the reasons Notified Bodies have refused applications
- Timeframes to CE Mark devices to the MDR and IVDR
Future Revisions of Dashboard
The dashboard currently only contains information from Notified Body surveys. However, it will eventually be updated to contain information from other stakeholders: manufacturers, authorized representatives, health service providers, patient representatives, and competent authorities.
If you would like to participate in the EU Commission’s survey, you may do so by clicking here. The survey deadline* is 31 January 2024.
*The EU Commission extended the response deadline on 15 January 2024. The deadline cutoff to respond was originally 15 January 2024.
Further Reading
You may be interested in the following resources:


