Today the ‘Manual on Borderline and Classification in the Community Regulatory Framework for Medical Devices (September 2022)’ was released.
It can be found: HERE
This Manual is not intended to replace other classification guidance documents, such as:
-
- MDCG 2022-5 – Guidance on borderline between medical devices and medicinal products
- MDCG 2021-24 – Guidance on classification of medical devices
- MDCG 2020-16, Rev.1 – Guidance on classification for IVDs
- MDCG 2019-11 – Qualification and classification of software
Instead, the Manual is intended to record the agreements reached by the Borderline and Classification Working Group (BCWG), following the exchanges under the Helsinki Procedure.
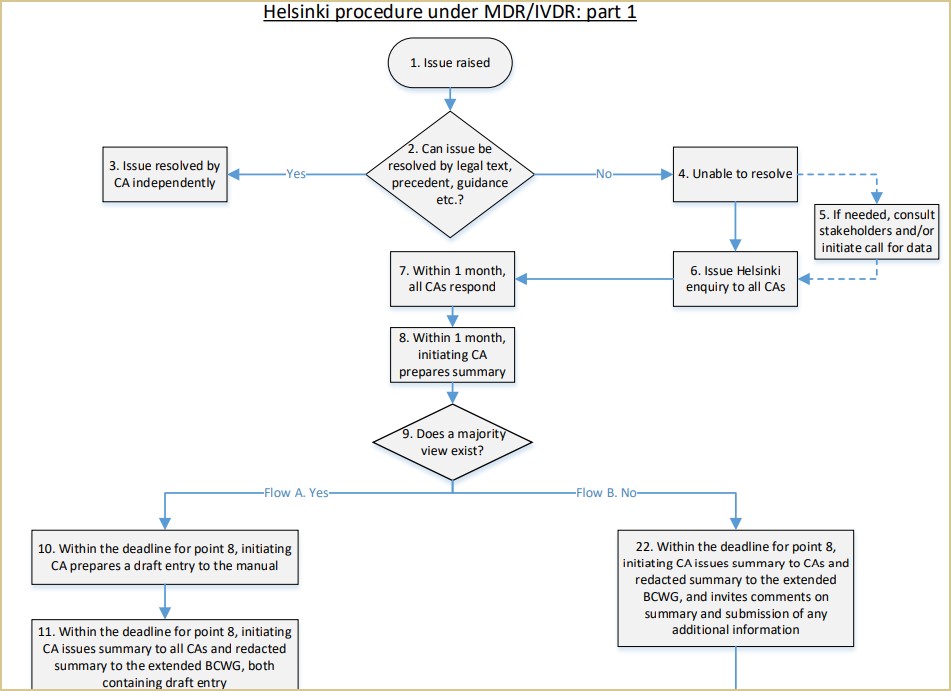
The Helsinki Procedure is a system ‘to allow consultation among competent authorities (CAs) on borderline and classification issues concerning medical devices and to ensure that appropriate guidance is published in the Manual on Borderline & Classification for Medical Devices…’
Below is an excerpt of a flowchart, to give a sense of the Helsinki Procedure process.